 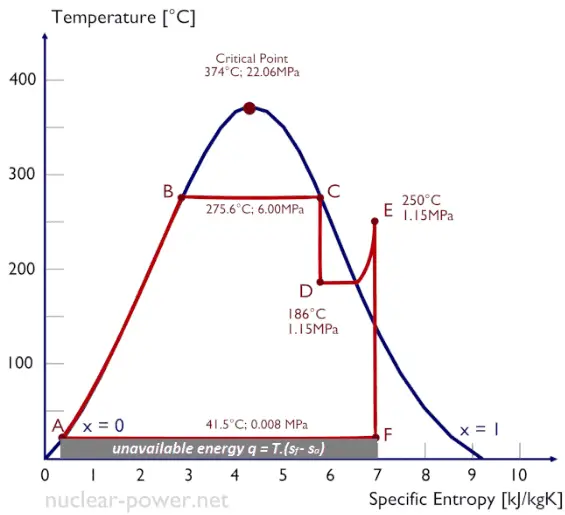 
This equation, which relates the microscopic details, or microstates, of the system (via W) to its macroscopic state (via the entropy S), is the key idea of statistical mechanics. Note that the exact definition of entropy is:Įntropy = (Boltzmann’s constant k) x logarithm of number of possible states This constant entropy is known as the residual entropy, which is the difference between a non-equilibrium state and crystal state of a substance close to absolute zero. glass) some finite entropy remains also at absolute zero, because the system’s microscopic structure (atom by atom) can be arranged in a different ways (W ≠ 1). 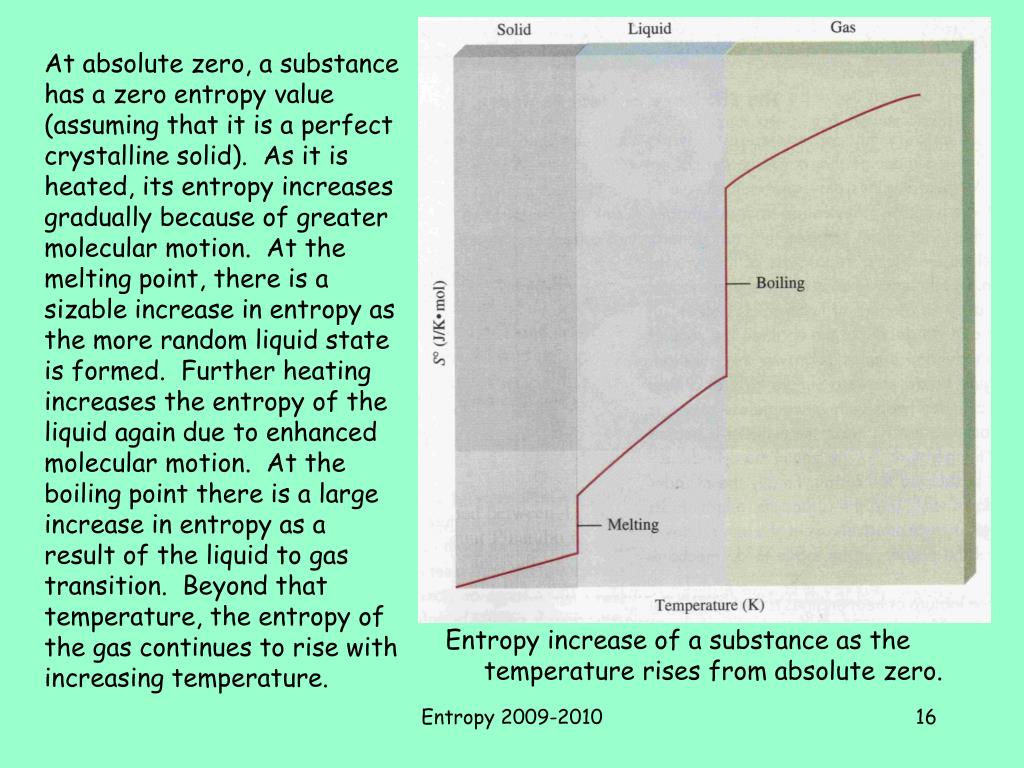
any amorphous solid) do not have a well-defined order at absolute zero. The Nernst heat theorem was later used by a German physicist Max Planck to define the third law of thermodynamics in terms of entropy and absolute zero. It is impossible for any process, no matter how idealized, to reduce the entropy of a system to its absolute-zero value in a finite number of operations. Nernst Heat Theorem (a consequence of the Third Law) is: Or conversely the absolute temperature of any pure crystalline substance in thermodynamic equilibrium approaches zero when the entropy approaches zero. As can be seen, the third law of thermodynamics states that the entropy of a system in thermodynamic equilibrium approaches zero as the temperature approaches zero. Therefore the third law of thermodynamics is often referred to as Nernst’s theorem or Nernst’s postulate. For this research Walther Nernst won the 1920 Nobel Prize in chemistry. The third law of thermodynamics was developed by the German chemist Walther Nernst during the years 1906–12. This allows us to define a zero point for the thermal energy of a body. The entropy of a system approaches a constant value as the temperature approaches absolute zero.īased on empirical evidence, this law states that the entropy of a pure crystalline substance is zero at the absolute zero of temperature, 0 K and that it is impossible by means of any process, no matter how idealized, to reduce the temperature of a system to absolute zero in a finite number of steps. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
